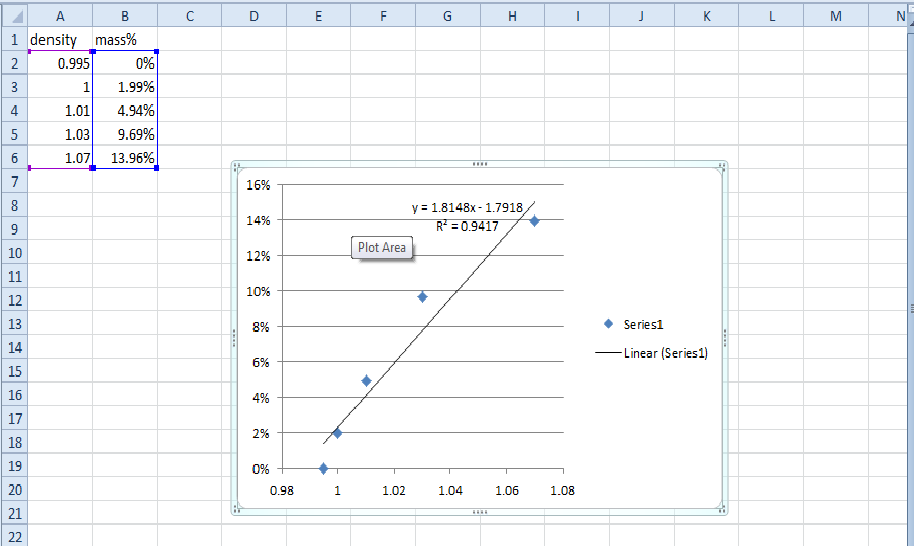
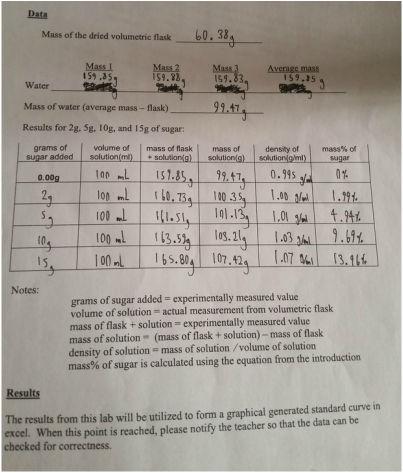
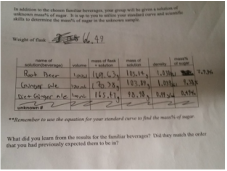
| On Thursday and Friday we had an amazingly exciting lab on sugar content. What we had set about to do was test substances to see how adding sugar affects the weight of them, and see if with the data collected, we could accurately predict how much sugar is contained in other mixtures. It was not the most exciting lab but it does show how research and repeated methods can lead to accurate hypotheses. What we did was measure out water and record it's weight when we added various amounts of sugar. From 2g to 15g, and using the formula 100(Mass sugar/Mass solution), we found what percent of the mass was sugar. Using this new information we were plugged in into an excel graph to see the pattern and acquire a equation for later. Next we chose three different beverages and repeated the process. The three we used for this experiment were: root beer, ginger ale, and diet ginger ale (all courtesy of Trader Joes). Just thinking abut the three sodas, we expected root bear to have the most sugar, followed by ginger ale, and lastly diet ginger ale. After doing the measurements, we plugged our data into our equation y = 1.8148x - 1.7918, and we were able to find the mass% of sugar quite easily. Upon doing so, we were surprised to find that ginger ale was much more sugary than root beer (diet of course was still last with 0.49%). Our experiment did not exactly follow our hypotheses but we were able to predict the amount of sugar in our beverages based on previous repeated data. Nothing much was to come as a result from the lab and therefore I feel like the lab went well in the sense that we completed what we had set out to do. I had the role of recording data and working out the other numbers through our formulas. I feel like I did contribute greatly to get the results that we did, because without the math then we couldn't have done the graph and equation to represent our data and help move us along to part two. I worked with Josh and Mira for this lab, they're are both great partners and I would gladly work with them again on another experiment. Although we did finish what needed to get done I feel like a lot of the work rested on me. With the information gathered I'm sure we could continue this experiment not only with other beverages, but maybe even doing different equations to predict other things such as the amount of an element present in a solution or the like. Somethings I took away from this was that with the right information you can easily prove and disprove anything. When using repeated experiments, it's very easy to trace a pattern and accurately predict the solution to your question. This also showed me how the periodic table was formed as scientists most likely did countless experiments with the elements and found those patterns that helped them predict what the missing spaces were. |
|
0 Comments
An old worksheet for measurement that involves significant digits. Many mistakes were made during this process as seen below. A take home quiz about atomic structure. We simply showed the atomic structure of different elements. A longer worksheet about the structure of atoms and the different parts that can be found within them. This one took much longer than most worksheets as we were describing each part of an atom and other subatomic particles. Another quiz about the periodic table and it's elements. This one was easier than the usual quiz but I actually had a lot of trouble with the last question. When researching the reactivity of elements, the closest I could find was sodium (Na) and magnesium (Mg). So I just chose the element that was still within the same group.
A short worksheet about different elements on the periodic table. Some things were a little tricky such as finding the number of valence electrons in the transitional metals, but all in all, a relatively easy one that once you know what to do, it's smooth sailing.
On Wednesday, we had a lab that involved hydrogen and atoms. Beforehand though, of course we had a short introduction to atoms. Atoms are made of three main subatomic particles, protons (p+), neutrons (n0), and electrons (e-). The neutrons and protons reside in the nucleus, or center, of the atom. The electrons orbit the nucleus at certain distances, staying within their shells. Neutrons and protons both have significant mass in an atom, meaning that an atoms weight mostly depends on the amount of protons and neutrons. We learned how when two atoms that have differing elections in their outermost shell are brought together, ones with less are more likely to react and give their electrons to those with more. In our lab we were looking to see how reactive hydrogen is.
For the actual experiment, we placed strips of magnesium in a volumetric flask then added some citric acid and water. The reaction was very quick so we placed a balloon over the flask to see the hydrogen build up in it. When the water came into contact with the magnesium and citric acid, it began to fizz, the glass started to heat up as I assume the hydrogen was being released from the reaction. After the magnesium was gone, we tied off the balloon and watched as it floated gently skywards. For stage two, we attached these balloons to meter sticks and placed the over a small candle while staying a ways back (about a meter away!). When the balloon came into contact with the flame a large explosion was all that was left of the balloon. From what we had previously learned, I assumed that the glass was getting hot because of the energy being released from the reaction. The balloon began to fill up with hydrogen that was being released from mixing the compounds. In the mixing I suppose that the hydrogen found in both citric acid and water was let go of when they came into contact with the magnesium. Because of hydrogen's high reactivity, when it the balloon began to heat up, it expanded very quickly, causing the explosion and was then burned up by the fire, explaining the left over (for lack of a better word) fire imprint. This was a very interesting experiment and I would love to do some more reactions in the future involving hydrogen. I say hydrogen because I know how reactive it is, so there must be a lot that we can do with it to. I was as surprised as everyone else when we were doing the exploding balloons, I figured it would pop, but I didn't expect it would be so loud/deep. It showed me how strong just a bit of hydrogen is when introduced to heat. |
AuthorHello there! I am a sophomore here at High Tech High, and on this blog I will be making weekly posts to what I've been doing in my 10th grade chemistry class! So check back often! Archives
May 2015
Categories |






 RSS Feed
RSS Feed
